Could this case of very early onset IBD actually be a primary immunodeficiency disorder?
A 3-month-old patient was hospitalized at Children’s Hospital Colorado for what appeared to be very early onset inflammatory bowel disease, or VEOIBD. During their pursuit to learn more, the team of immunologists and gastroenterologists uncovered a novel genetic defect.
Pediatric gastroenterologist Edwin de Zoeten, MD, PhD, was the initial specialist on the case.
Along with common IBD symptoms like diarrhea, blood in the stool and failure to thrive, Dr. de Zoeten and his team found marked nodularity in the esophagus, along with lymphoid inflammation in the stomach and intestine. Additional evaluation revealed cytomegalovirus, or CMV, in the intestines and blood, and a myriad of other symptoms including a persistent dermatitis.
Although technically a pediatric gastroenterologist, Dr. de Zoeten has a PhD in immunology. So when the typical treatment methods for IBD weren’t helping, he knew the problem was likely more complicated.
“Patients who present like this can have a monogenic disease, or a single gene that’s causing their intestinal inflammation,” he says. “We consulted with our immunology team, and we collectively recommended whole exome sequencing.”
Identifying the novel genetic defect
Whole exome sequencing revealed a homozygous mutation in the IL2RB gene, encoding a protein called interleukin-2 receptor subunit beta, or IL-2Rß. With features suggesting the mutation was pathogenic, Cullen Dutmer, MD, pediatric immunologist and Medical Director of the Clinical Immunology Program, suggested looking at the mutation more closely.
“The IL-2 receptor has three subunits,” he says. “We know a lot about subunits alpha and gamma, including how defects within these subunits result in disease. But this is the first for subunit beta.”
The team discovered that for this particular patient, the gene mutation was disrupting the function of the protein. Further still, IL-2Rß is a subunit shared with another interleukin receptor called the IL-15 receptor. That only amplifies the problem, because cells that either express the IL-2 receptor or the IL-15 receptor are then affected. The cells of greatest interest were T cells and NK cells, whose hampered functionality was manifesting as multisystem autoimmune disease and susceptibility to CMV infection.
Due to the severity of the disease, the best course of treatment for the patient was a hematopoietic stem cell transplantation, also known as a bone marrow transplant. Although it was first used to treat some types of cancer, it’s now widely used for an assortment of immune system disorders.
The transplant resulted in a complete resolution of the patient’s symptoms, validating it as a likely curative treatment for some patients with IL-2Rß defects.
But what about therapies prior to — or instead of — transplant? Pediatric immunologist Elena Hsieh, MD, is working to learn more.
Studying IL-2Rß in the lab
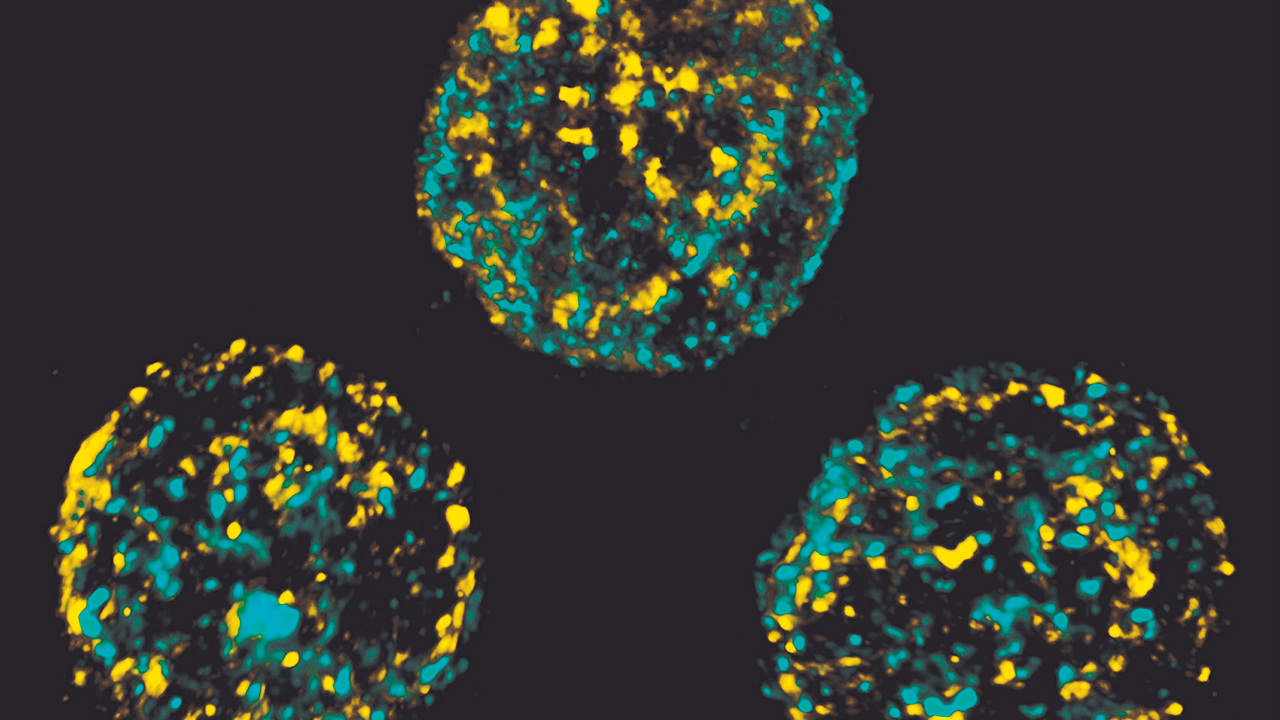
In her lab here on the Anschutz Medical Campus, Dr. Hsieh is researching the underlying mechanisms of the published human IL-2Rß defect to understand how defects like these result in both the immunological and clinical presentations. To do so, she developed a mouse model with a homologous murine mutation that recapitulates the human disease, and she is currently studying several different aspects of the disease.
She’s assessing cellular function by isolating NK cells from a wild type mouse and an IL2RB mutant mouse and evaluating how those cells interact with target cells. Can the cells from the mutant mouse kill effectively? If not, why not? Furthermore, she’s evaluating the ability of mutant cytotoxic T cells to respond to IL-2 and IL-15, while also looking at transcriptional programming of the cells through RNA sequencing.
“It’s true that these patients have an abnormal receptor, but they also have abnormal serum cytokine levels,” says Dr. Hsieh. “The abnormalities we see at the cellular level could be a product of the abnormal receptor, the abnormal cytokine environment, or a combination of both.”
She’s taking the mutant cells and putting them in a healthy mouse environment, and she’s proposing a hypothesis: If you correct the environment in which this mutant cell develops and matures, can you correct the NK cell defect?
If they can pin significant effects on the cytokine environment, that’s something they can modify.
“There are several new medications that are designed to block cytokines, their receptors, or their downstream signaling pathways,” says Dr. Hsieh. “If we could show that correcting the environment actually corrects much of the disease, we have an avenue to apply therapies that already exist to help treat the disease.”
The paper “A novel human IL2RB mutation results in T and NK cell-driven immune dysregulation” was published in the Journal of Experimental Medicine in June 2019.
Jeffrey Modell Diagnostic and Research Center for Primary Immunodeficiencies
In addition to work specific to the IL2RB mutation, Children’s Colorado is bolstering its clinical programs to better identify patients who may have these types of diseases and offer more comprehensive diagnostic evaluations and care.
“The technology to diagnose these patients didn’t really exist 20 years ago,” says Dr. de Zoeten. “Fifteen years ago, it would have been in its infancy, and even 10 years ago it was still really expensive. Because of that, many cases across the country were likely misdiagnosed or missed.”
Now, with a multidisciplinary team that includes immunologists, rheumatologists, gastroenterologists, pulmonologists, hematologists, oncologists and bone marrow transplant specialists — plus a budding genomics program — Children’s Colorado was recently designated a Jeffrey Modell Diagnostic and Research Center for Primary Immunodeficiencies.
“The designation underscores the quality of clinical care patients with suspected and known primary immunodeficiency disorders receive at Children’s Colorado and signifies the immunology department as a leader in research,” says Dr. de Zoeten.
Featured Researchers

Edwin de Zoeten, MD, PhD
Pediatric gastroenterologist
Children's Hospital Colorado

Elena Hsieh, MD
Immunologist
Allergy and Immunology Center
Children's Hospital Colorado
Associate professor
Pediatrics–Immunology and Microbiology
University of Colorado School of Medicine





 720-777-0123
720-777-0123