Thanks to advancements in pharmaceuticals during the last 25 years, a variety of medications treat seizures in children. Unfortunately, a third of those children continue having seizures anyway.
"We can only get so far with epilepsy research using non-specific approaches,' says Amy Brooks-Kayal, MD, Chief of Pediatric Neurology. As a result, her team's research efforts are getting increasingly specific.
Homing in on focal cortical dysplasia
Focal cortical dysplasia, or FCD, is a malformation of cortical development caused by a genetic mutation that occurs in utero, leading to disordered layers of neurons in the cerebral cortex. It's also a common cause of drug-resistant epilepsy in children.
"Patients with this diagnosis often require resection surgery for treatment of their epilepsy," says pediatric neurosurgeon Allyson Alexander, MD, PhD. "Unfortunately, this surgery is invasive and not always successful. Also, some patients are not candidates for resection surgery due to the location of the dysplasia in their cerebral cortex."
Epileptologists and neurosurgeons know that FCD leads to epilepsy, but they're still exploring what specific aspects of the disorder cause seizures. That work has led to the discovery of somatic mutations in several genes of what's called the mammalian target of rapamycin pathway, or mTOR.
"This pathway regulates cell metabolism, growth, proliferation and survival," says Dr. Alexander. "Now we want to understand how genetic and histological changes in FCD lead to a brain network that generates difficult-to-control seizures."
The abnormal bursting patterns of FCD
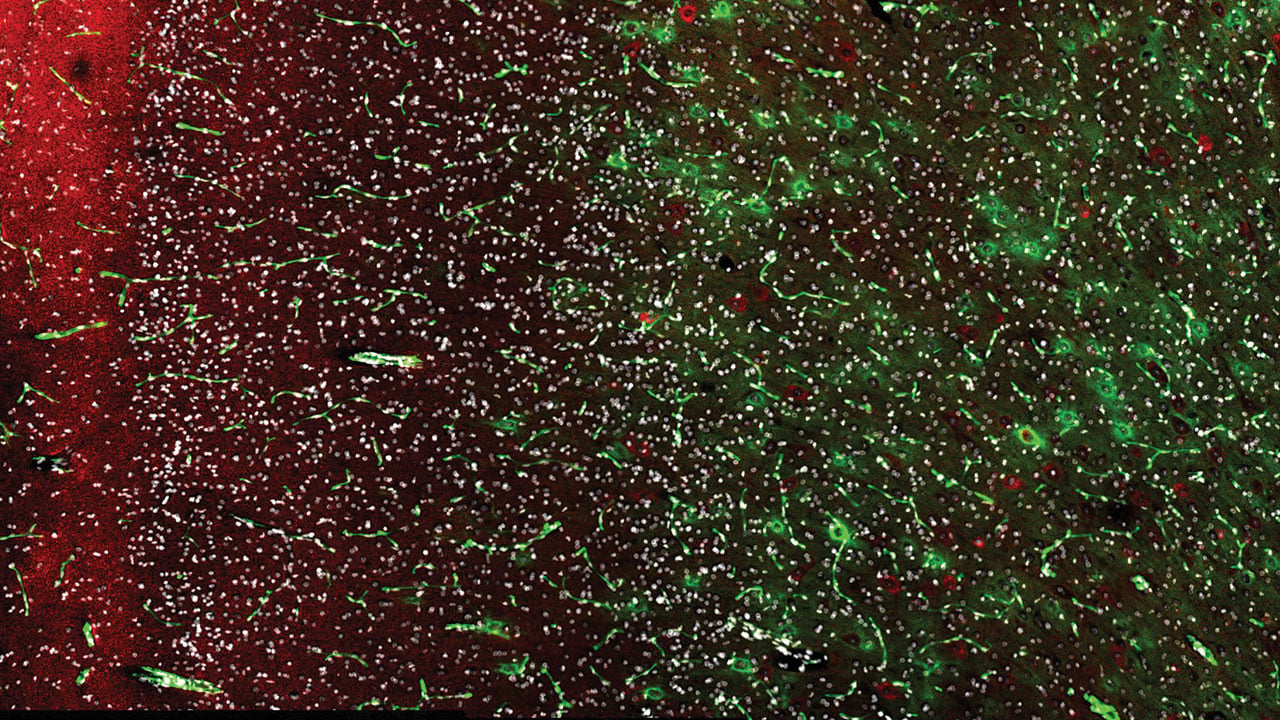
One potential clue is an abnormal bursting pattern in some brain cells of patients with FCD, discovered by Molly Huntsman, PhD, a researcher with the University of Colorado's Skaggs School of Pharmacy, just across the Anschutz Medical Campus from Children's Hospital Colorado. These bursting patterns have proven to promote seizure-like activity in rodent and computational models.
"We want to understand how genetic and histological changes in FCD lead to a brain network that generates difficult-to-control seizures."
- ALLYSON ALEXANDER, MD, PHD
Based on these findings, Dr. Alexander is using a two-pronged approach — using resected human brain tissue and an experimental FCD mouse model — to further this research. She's hoping to determine whether neocortical pyramid cells exhibit abnormal bursting behavior, and whether this burst firing pattern is due to alterations in the expressions of T-type calcium channels.
Digging deeper for targeted focal cortical dysplasia treatment options
Discovering T-type calcium channels as the culprit for FCD seizures would be good news for patients, as the channel is a known target for ethosuximide, an FDA-approved drug used to treat some forms of epilepsy.
"As one part of this research, we're taking tissue from FCD patients resected during surgery, slicing it very thin and putting it into an oxygenated environment," says Dr. Alexander. "This allows us to keep the neurons alive and record their activity using a micro-pipette."
However, this approach has limitations due to the lack of normal human brain tissue available, which is why Dr. Alexander's team is also developing a mouse model for exploratory research. Based on a previous model, it uses in-utero electroporation to modulate the mTOR pathway in a subset of neurons, leading to an area of disordered cortical layers similar to that seen in patients with FCD.
Once the model is up and running, Dr. Alexander will study the bursting cells to learn whether the T-type calcium currents play a role in the bursting behavior. If so, her team will test whether the T-type calcium blocker could stop these seizures.
"If we're going to move beyond where we are today, we have to better understand, and then specifically target, the underlying mechanisms that cause different types of epilepsy," says Dr. Brooks-Kayal. "We're thinking beyond seizures."
Featured Researchers

Allyson Alexander, MD, PhD
Pediatric neurosurgeon
The Neuroscience Institute
Children's Hospital Colorado
Assistant professor
Neurosurgery-Peds
University of Colorado School of Medicine

Amy Brooks-Kayal, MD
Pediatric neurologist
Pediatric Neurology
Children's Hospital Colorado
Molly Huntsman, PhD
Associate professor
School of Pharmacy
University of Colorado School of Medicine





 720-777-0123
720-777-0123