Key takeaways
-
Our study is the largest to research airway microbiota from bronchoalveolar lavage fluid (BALF) across age and disease spectrum in CF.
-
CF microbiota detected in BALF differs with patient age.
-
In CF patients under 2 years old, non-traditional taxa predominate.
-
Microbial diversity and relative abundance of non-traditional taxa (Streptococcus, Prevotella and Veillonella) were inversely associated with airway inflammation.
Cystic fibrosis (CF) is a genetic disease that causes persistent lung infections, making it difficult for patients to breathe. According to the Cystic Fibrosis Foundation, more than 30,000 people in the United States — and more than 70,000 people worldwide — are living with the disease.
Research background: Detecting cystic fibrosis airway microbiota through bronchoscopy with bronchoalveolar lavage fluid (BALF)
In the lungs of patients with CF, mucus clogs the airways and traps bacteria. The pathogens most frequently cultured from CF airway samples are Pseudomonas aeruginosa and Staphylococcus aureus. These "traditional" CF bacteria are associated with chronic airway infection, inflammation and pulmonary decline.
Recent molecular DNA sequencing efforts have revealed that bacterial communities in CF airways are more complex, but most studies have evaluated sputum (coughed-up mucus), which contains a mix of lower and upper airway bacteria. Bronchoscopy with bronchoalveolar lavage fluid (BALF) collection is preferred for studying lower airway bacteria with minimal upper airway contamination, but routine surveillance bronchoscopy is not recommended for CF patients.
Defining bacterial communities in CF — and distinguishing pathogenic bacteria that cause disease from commensal (not harmful) bacteria — is important for developing new treatment strategies for CF.
Edith Zemanick, MD, and other researchers at the Mike Morris Cystic Fibrosis Research and Care Center in the Breathing Institute at Children's Hospital Colorado, led a multi-center study to evaluate the CF airway microbiome in BALF samples from CF patients of all ages and disease severity.
Research methods: Analyzing bacterial DNA in airway samples from patients with CF and control patients
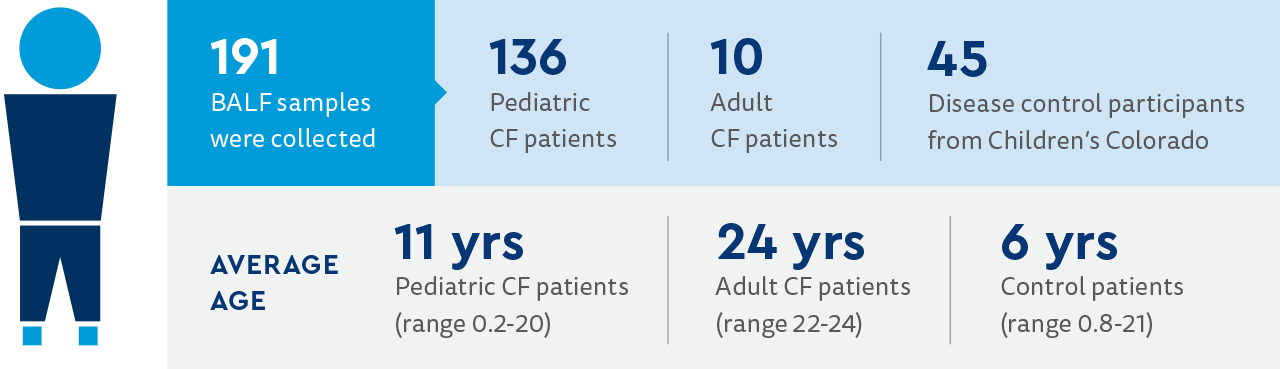
The following is a summary of the study participants and procedures of the study.
Study participants:
- Patients with CF were from 13 centers in the U.S.
- Patients were recruited at the time of clinically indicated bronchoscopy.
- Patients with CF were ages 2 months to 42 years.
- Disease controls were pediatric patients undergoing bronchoscopy for indications other than CF.
- Disease control patients came from Children's Colorado.
- CF patients were categorized as:
- Clinically stable (no reported respiratory symptoms)
- Pulmonary exacerbation (respiratory symptoms leading to bronchoscopy)
- Unknown
Study procedures:
- Bronchoscopy/BALF collection followed standard clinical procedure to minimize upper airway contamination.
- Standard bacterial cultures were performed by each center's clinical microbiology laboratories.
- BALF samples were frozen and shipped overnight to Children's Colorado.
- DNA extractions were performed at study site.
- Total bacterial load was measured at study site.
- Bacterial profiles were determined by molecular DNA sequencing.
Research results: CF airway microbiome varies by age and airway inflammation
The most common indications for bronchoscopy in CF patients were persistent cough (41%), unresponsiveness to treatment (29%), need for lower airway culture (27%), concurrent surgery (21%) and lung function decline (8%).
The most common indications for bronchoscopy in disease control patients were asthma or wheezing (38%), pneumonia/recurrent pneumonia (36%), dysphagia/esophageal reflux (33%) and chronic cough (24%).
Bacterial cultures, total bacterial load and bacterial types detected in BAL fluid
BALF bacterial cultures were positive in 90% of adult CF, 70% of pediatric CF and 33% of disease control participants. The total bacterial load (TBL) was higher in pediatric CF compared with disease control samples. TBL did not differ between adult and pediatric CF. Bacterial populations were more diverse in disease control compared with CF participants.
Traditional CF pathogens (Pseudomonas, Staphylococcus, Stenotrophomonas, Haemophilus, Achromobacter and Burkholderia) were the dominant bacterial populations in 47% of pediatric CF and 60% of adult CF BALF samples. None of these bacterial populations were dominant in disease control samples.
Interestingly, 20% of pediatric and adult CF BALF contained a dominant bacterium with an unknown association to disease severity and progression in CF, which the authors termed "non-traditional" (Streptococcus, Prevotella, Bordetella, Veillonella, Moraxella, Neisseria and Corynebacterium).
Relationship between cystic fibrosis bacterial communities, age and clinical characteristics
TBL did not differ across CF age groups, but bacterial diversity was higher in younger age groups. The non-traditional bacterium Streptococcus was most prominent in young children with CF, and traditional CF bacteria comprised less than 50% of the bacterial community in children under age 2. By age 6, a median of 70% of the bacterial communities consisted of traditional CF pathogens.
Non-traditional bacteria (Streptococcus, Veillonella and Prevotella) were associated with decreased markers of airway inflammation. Pseudomonas aeruginosa abundance and markers of inflammation were higher in CF patients with pulmonary exacerbation compared with clinically stable participants.
Research discussion: Traditional and non-traditional bacteria in the CF airway microbiome
This large, multi-center study demonstrated that lower airway bacteria in patients with CF differ by patient age. Non-traditional bacteria (Streptococcus, Veillonella, Prevotella and Haemophilus) were more abundant in younger children and decreased in abundance in older patients. Bacterial diversity in younger patients was associated with less airway inflammation.
Traditional CF bacteria (Pseudomonas) became increasingly abundant as patients got older. Bacterial diversity was lower in older CF patients, due to the prominence of Pseudomonas, and airway inflammation increased. Types of bacteria did not differ between CF patients who were clinically stable and those who had respiratory symptoms.
Research conclusion: Investigating the role of non-traditional cystic fibrosis bacteria
The clinical impact of non-traditional bacteria in CF is still under investigation. These bacteria may be less pro-inflammatory than traditional CF bacteria. Understanding the diverse non-traditional bacterial communities in young CF patients and how those communities change as traditional CF bacteria become more abundant may improve early treatments.
Refer a patient
The Mike McMorris Cystic Fibrosis Research and Care Center in the Breathing Institute at Children's Colorado is one of the premiere cystic fibrosis (CF) clinical care and research centers in the country. Contact us at 720-777-6181 or cysticfibrosis@childrenscolorado.org.
Featured Researchers

Edith Zemanick, MD
Associate Director of the Cystic Fibrosis Research Center
Children's Hospital Colorado
Professor
Pulmonary Medicine - Department of Pediatrics
University of Colorado





 720-777-0123
720-777-0123